Peptides & Peptide Synthesis Products
Boc-Val-OH
Catalog No: 48125
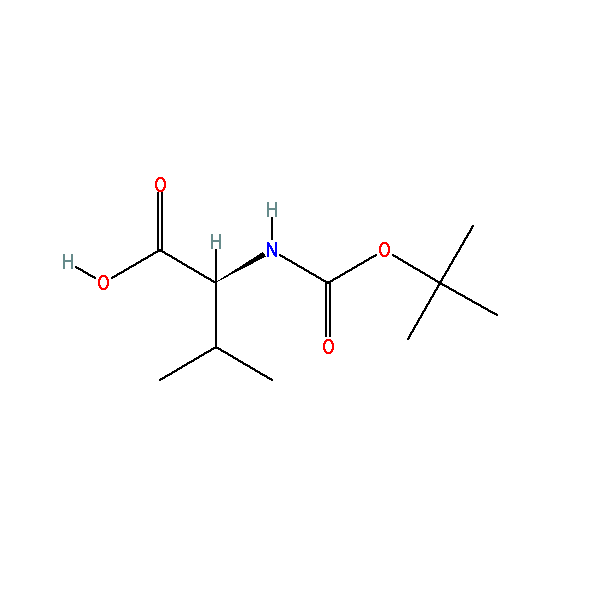
(2S)-3-methyl-2-[(2-methylpropan-2-yl)oxycarbonylamino]butanoic acid, CAS: 13734-41-3, MW: 217.26, Formula: C10H19NO4
(2S)-3-methyl-2-[(2-methylpropan-2-yl)oxycarbonylamino]butanoic acid, CAS: 13734-41-3, MW: 217.26, Formula: C10H19NO4
$15.00
SKU
48125
(2S)-3-methyl-2-[(2-methylpropan-2-yl)oxycarbonylamino]butanoic acid
| Catalog Number | 48125 |
|---|---|
| CAS | 13734-41-3 |
| M.W. | 217.26 |
| Formula | C10H19NO4 |
| IUPAC Name | (2S)-3-methyl-2-[(2-methylpropan-2-yl)oxycarbonylamino]butanoic acid |
| Also Known As |
|
| InChIKey | SZXBQTSZISFIAO-ZETCQYMHSA-N |
| InChI | InChI=1S/C10H19NO4/c1-6(2)7(8(12)13)11-9(14)15-10(3,4)5/h6-7H,1-5H3,(H,11,14)(H,12,13)/t7-/m0/s1 |
| SMILES | CC(C)[C@@H](C(=O)O)NC(=O)OC(C)(C)C |
Write Your Own Review